AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
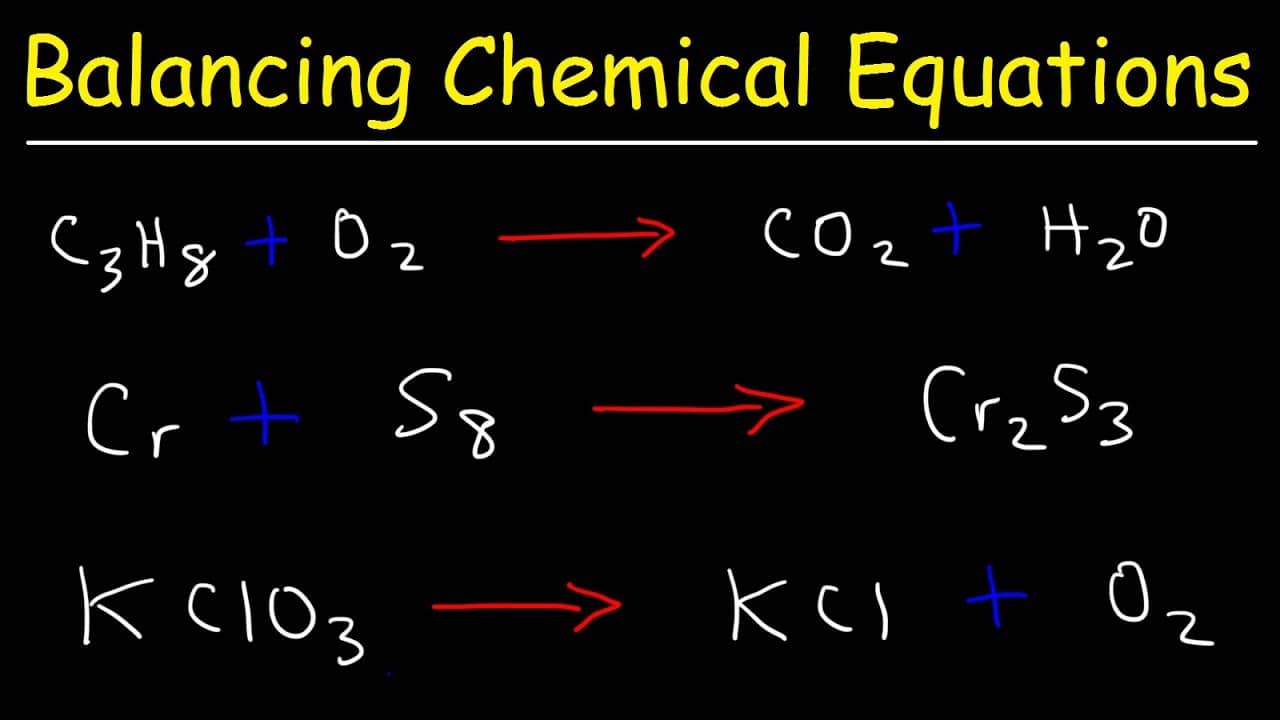
Assuming this is true, the reaction would be quite spectacular: an aluminum atom would appear out of nowhere, and two (2) iron atoms and one (1) oxygen atom would magically disappear. $$Al + Fe_3O_4\longrightarrow Al_2O_3 + Fe$$īecause no coefficients are mentioned for any of the terms, we can assume that one (1) mole of Al reacts with one (1) mole of Fe3O4 to produce one (1) mole of Al2O3. Such is the case when the equations are not balanced properly. There are times, however, when we have to do some work before we can use the coefficients of the terms to determine the relative number of molecules of each compound. Rectangular To Polar Calculator Balancing Chemical Equations In other words, 40 grams of calcium equals one mole, 80 grams equals two moles, etc. Based on the atomic or molecular mass of a substance, a mole of that substance is equal to a gram of the substance.Ĭalcium, for example, has an atomic mass of 40. 
The conversion can be done easily when the atomic and/or molecular mass of the substance(s) is known. It is often necessary to convert between moles and grams of a substance. Therefore, you have the same number of moles of AgNO3, NaCl, AgCl, NaNO3. Since there are no numbers in front of the terms in the equation above, each coefficient is assumed to be one (1). Likewise, if you have a mole of carrots, you have 6.022 x 1023 carrots. When you have a dozen carrots, you have twelve of them. A mole simply represents Avogadro’s number (6.022 x 1023) of molecules. The following graphic illustrates many of the concepts described above: The Moleįrom the equation above, we can determine the number of moles of reactants and products. The notation above and below the arrows in the graphic below shows that this reaction must occur at a chemical Fe2O3, a temperature of 1000☌, and a pressure of 500 atmospheres. By providing information such as a temperature value, we can determine what conditions are needed for a reaction to take place. It is not uncommon for a variety of information to be written above or below the arrows. One (1) is assumed in the absence of a coefficient. An amount can represent either the relative number of molecules or the relative number of moles (described below). In all chemical equations, coefficients are used to indicate the relative amounts of each substance. Lastly, the (g) sign indicates that the compound is a gas. The (aq) sign stands for aqueous in water and means the compound is dissolved in water. 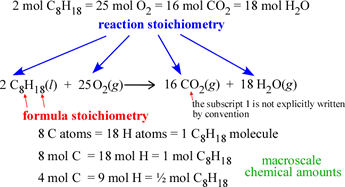
The (l) sign indicates that the substance is liquid. The (s) sign indicates that the compound is solid. There are often chemical equations that show what state each substance is in. By undergoing a chemical reaction, they are fundamentally changed. According to the equation, the reactants (AgNO3 and NaCl) react through some process to form products (AgCl and NaNO3). $$AgNO_3(aq) + NaCl(aq)\longrightarrow AgCl(s) + NaNO_3(aq)$$ĪgNO3 and NaCl are mixed in this equation. Nomenclature, or naming schemes, should have been covered in earlier readings.Ĭhemical equations are expressions of chemical processes. In this case, chlorine is called “chloride” because of its connection to sodium. We would use the notation “NaCl” to represent a molecule of table salt, sodium chloride, since “Na” stands for sodium and “Cl” stands for chlorine. As an example, the symbol “C” represents an atom of carbon, and “H” represents an atom of hydrogen. It is important to become familiar with these basic symbols in order to be successful in chemistry. To represent the various chemicals in chemistry, we use symbols. Stoichiometry Calculatorįor stoichiometry to be useful in running calculations about chemical reactions, a basic understanding of the relationships between products and reactants, as well as their reasons, is needed, which entails learning how reactions are balanced. Stoichiometry literally translates as the measurement of elements, since stoikhein means element and metron means to measure. Stoichiometry is a section of chemistry that uses relationships between reactants and/or products of a chemical reaction to determine desired quantitative data.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
